 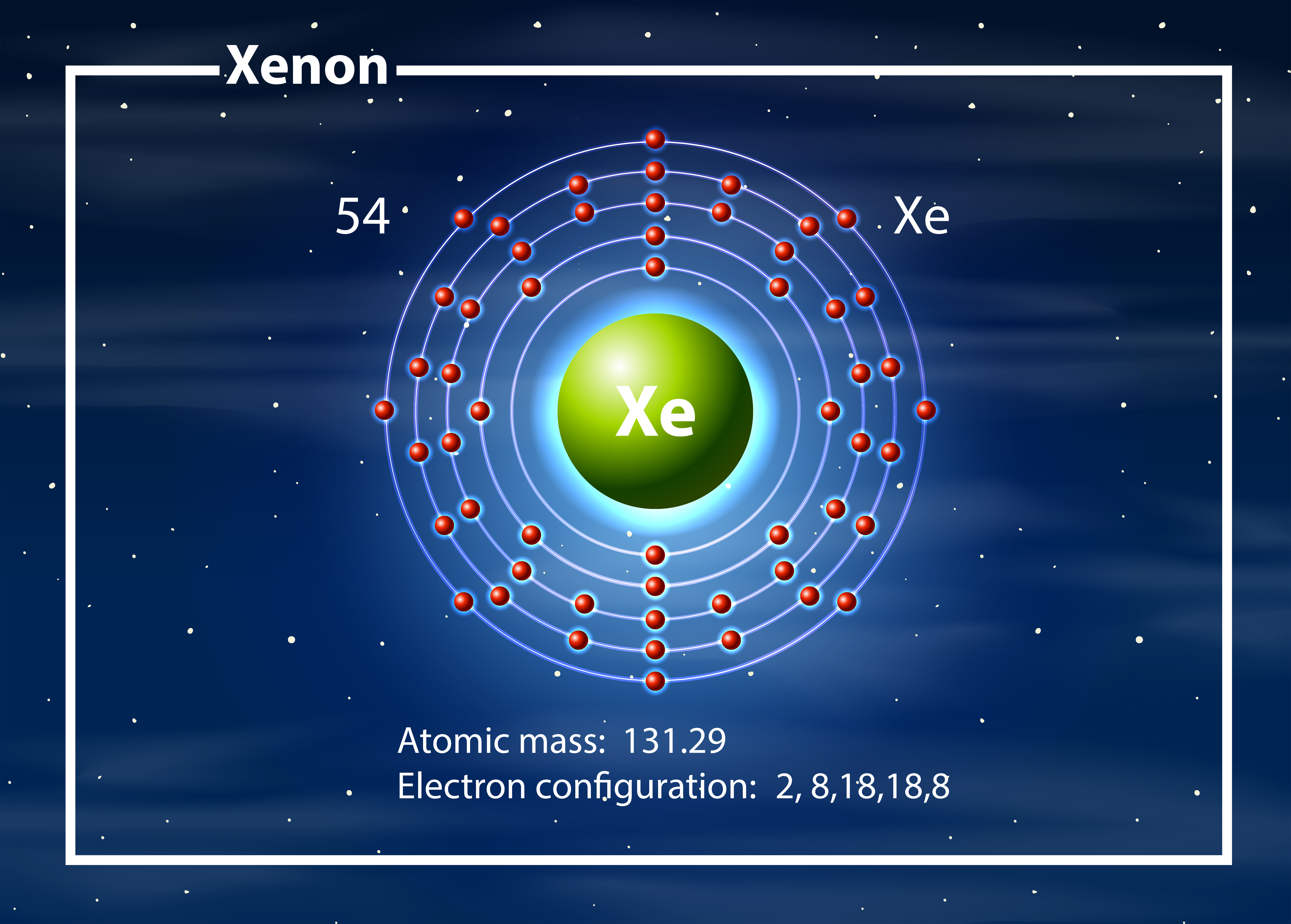 
For example, the electron configuration of the neon atom is 1s 2 2s 2 2p 6, meaning that the 1s, 2s and 2p subshells are occupied by 2, 2 and 6 electrons respectively.Įlectronic configurations describe each electron as moving independently in an orbital, in an average field created by all other orbitals. The hybridisation of XeF 4 is sp 3d 2.In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. The electronic configuration of xenon is 4d 10 5s 2 5p 6, and the ground state electronic configuration is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6. The key while writing this type of configuration is to find the nearest noble gas. For xenon, the nearest noble gas is Krypton (Kr), having configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6. The condensed electronic configuration of xenon is 4d 10 5s 2 5p 6. The hybridization of XeF 4 is sp 3d 2 XeF 4 Electron Configuration Xenon Condensed Electron Configuration.The electronic configuration of F is 1s 2 2s 2 2p 6.The electronic configuration of Xe is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 4 5d 2.Ground State Xenon Orbital Diagram XeF4 Electron Configuration The ground state orbital diagram is formed as per the ground state electronic configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6. Excited States of Xenon Electron Configuration Ground State Xenon Orbital Diagram The step-by-step formation of these excited states is explained with the help of the diagram given below. The excited state electronic configuration of xenon is given as: Excited State of Xenon Electron Configuration The ground state electronic configuration of xenon is: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 where 4s is filled before 3d and 5s is filled before 4d in ground state electronic configuration. Ground State Xenon Electron Configuration The unabbreviated electronic configuration of xenon is: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6. Xenon Unabbreviated Electron Configuration The electron configuration of xenon is notated as: 4d 10 5s 2 5p 6 where Kr has an electronic configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6. Xenon Electron Configuration Diagram Xenon Electron Configuration Notation In the electronic configuration of xenon is 4d 10 5s 2 5p 6 where the K shell (n=1) has 2 electrons, the L shell (n=2) has 8 electrons, the M shell (n=3) has 18 electrons, the N shell (n=4) has 18 electrons, and the O shell (n=5) has 8 electrons. The electron configuration diagram of xenon is given below. The 4p and 5s orbitals are filled with 6 and 2 electrons, respectively, followed by 10 in the 4d and 6 in the 5p orbital, making it a total of 54 electrons.The 3d orbital is filled with 10 electrons, which can have a maximum 10 electrons.Then, 3s, 3p and 4s orbitals are further filled with 2, 6, and 2 electrons respectively.Next, two electrons enter the 2s orbital, followed by 6 electrons in the 2p orbital.The first two electrons enter the 1s orbital, which can have a maximum two electrons.The step-by-step approach to writing the electronic configuration as per Aufbau principle is as follows: The electronic configuration of Xenon is 4d 10 5s 2 5p 6. How to Write Xenon Electron Configuration It will also discuss the compound XeF 4 and so much more. This article will cover the ground state and the excited state electronic configuration of xenon. It has an atomic number of 54, and electrons are arranged in atomic orbitals to attain an electronic configuration. It can be found in group 18, and it has an atomic symbol, ‘Xe,’ and an atomic mass of 131.293 u. 
Xenon has an electronic configuration of 4d 10 5s 2 5p 6. Let us learn about the electronic configuration of the xenon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
